If you could track only one KPI in a QC lab – what would it be?
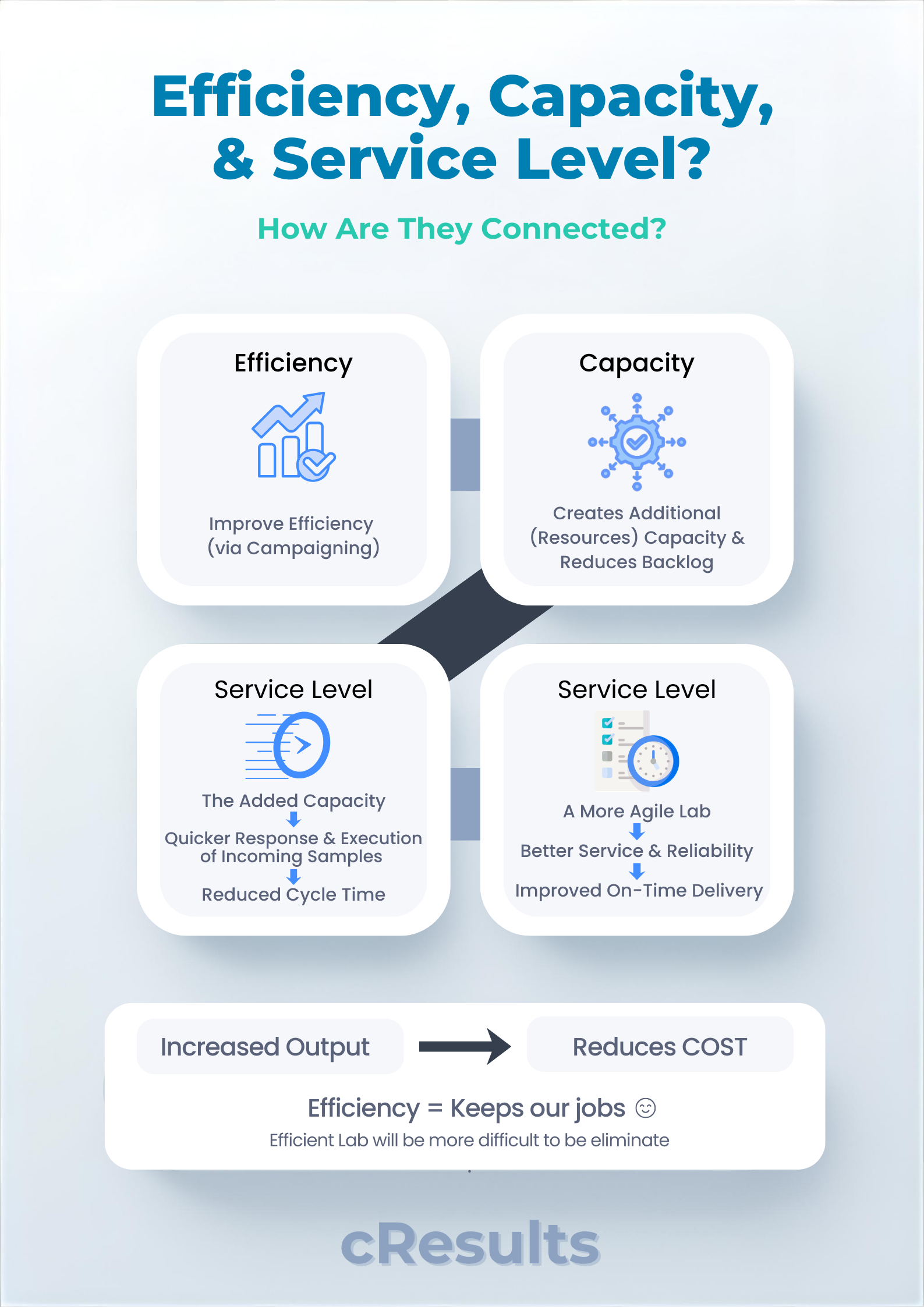
In our benchmark across 100 lab locations, most organizations monitor 6 KPIs spanning service level, efficiency, and right-first-time.
But one metric consistently reflects overall operational health:
Cycle Time.
When cycle time meets target consistently, it typically signals:
- Efficient analytical execution
- Scheduling aligned with batch release priorities
- Controlled OOS and minimal re-testing
- Reliable consumables and reagent availability
- Campaigning that improves throughput – not delays it
Cycle time is not just a performance metric.
It is a composite indicator of operational discipline, cross-functional coordination, and execution maturity across the QC organization.
The real question is: Are you managing cycle time proactively – or reacting to variability?
QC leaders looking to build a proactive approach to QC cycle time are welcome to contact us at Smart-QC to learn more.
