
Batch Record Review: Reduce Release Cycle Time with Smart-QA
How many hours does your batch sit “ready”… but not released? In pharma manufacturing, the final batch record review is often the…

How many hours does your batch sit “ready”… but not released? In pharma manufacturing, the final batch record review is often the…
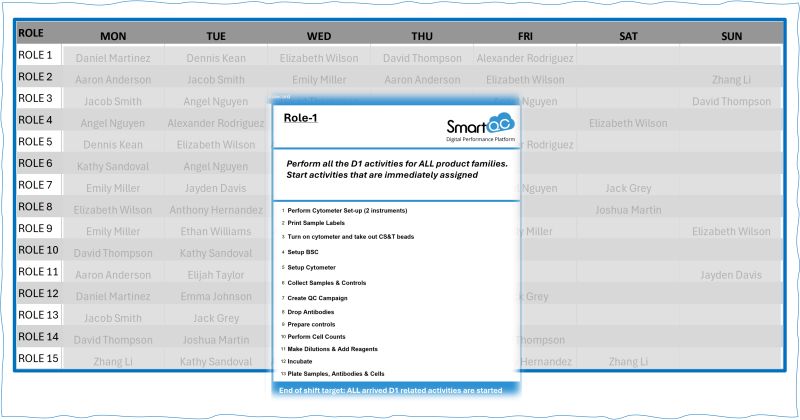
The key objective of QC laboratory role cards is to clearly define roles in alignment with the expected hands-on time and the…
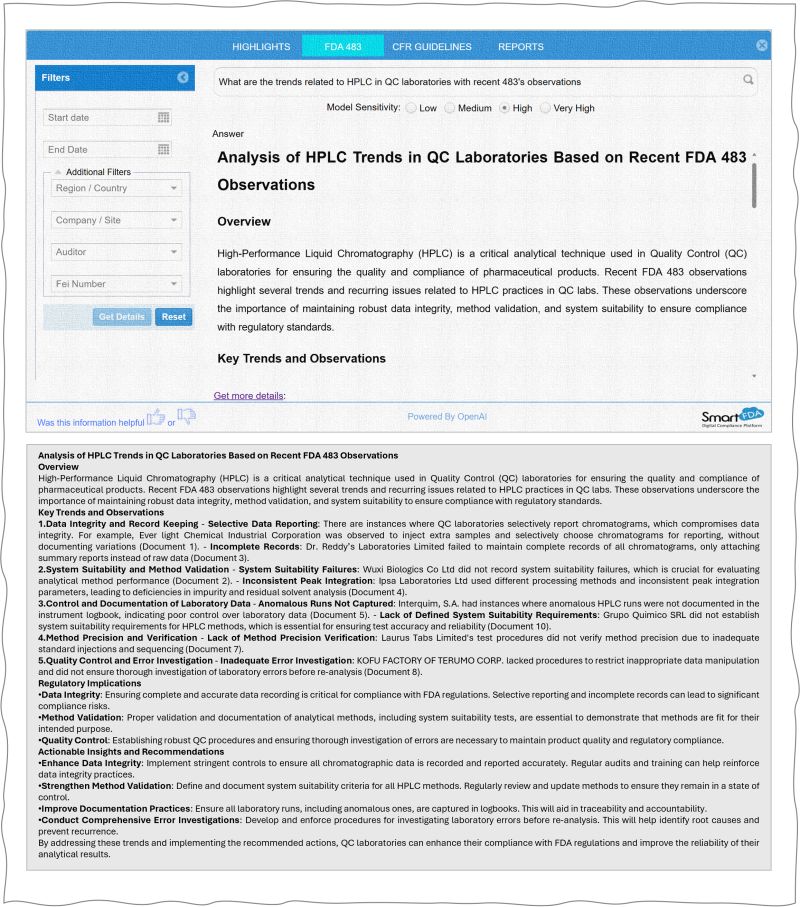
AI in the Compliance Space: How Smart-FDA Is Transforming FDA Compliance

Generative AI FDA Compliance: Smart-FDA Webinar Recap Generative AI FDA compliance is transforming how pharmaceutical and life sciences organizations manage regulatory risk…
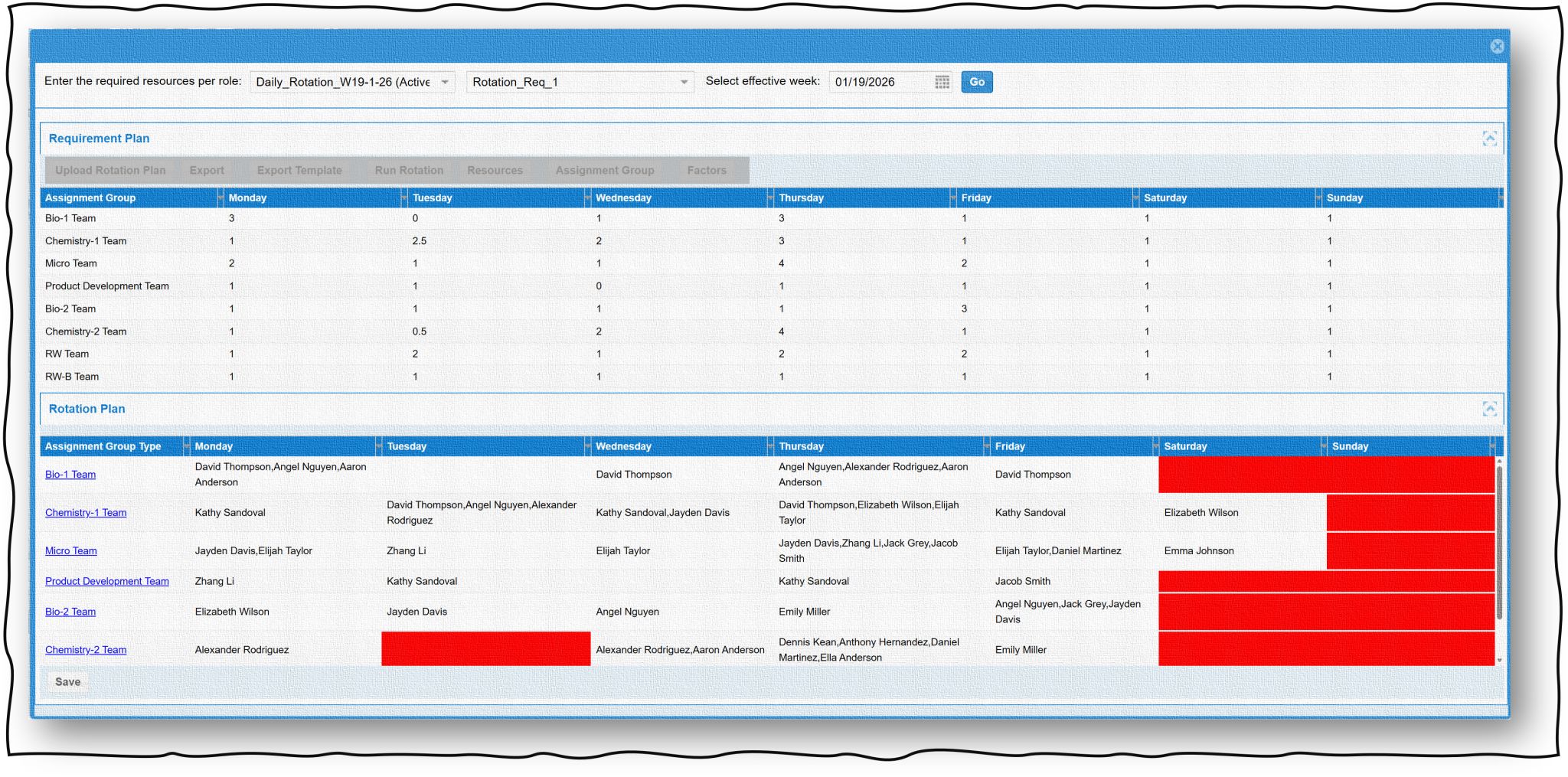
QC Laboratory Rotation Roles: 5 Smart Ways to Automate Scheduling Automating QC laboratory rotation roles is one of the most impactfulimprovements a…
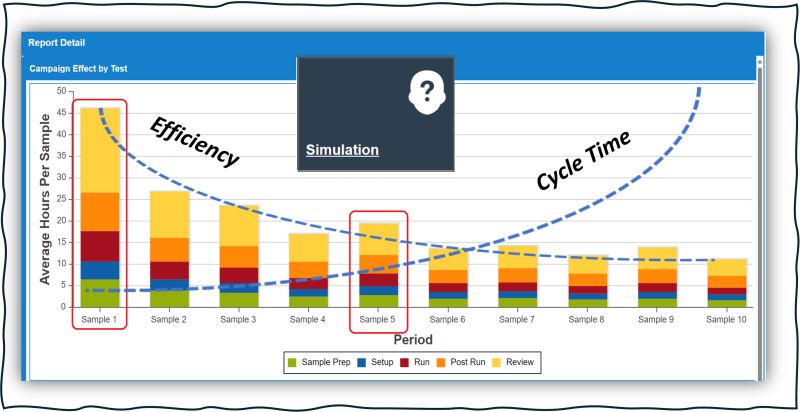
7 Proven Strategies to Improve Lab Efficiency and Cycle Time The Hidden Conflict Between Lab Efficiency and Cycle Time Lab efficiency and…