AI-Driven Compliance and CAPA FDA 483 Trends
AI is transforming compliance and with Smart-FDA, we’re helping lead the way. Every day, we ask Smart-FDA to analyze trends across thousands of FDA 483 observations so our customers can move from reactive compliance to proactive quality intelligence.
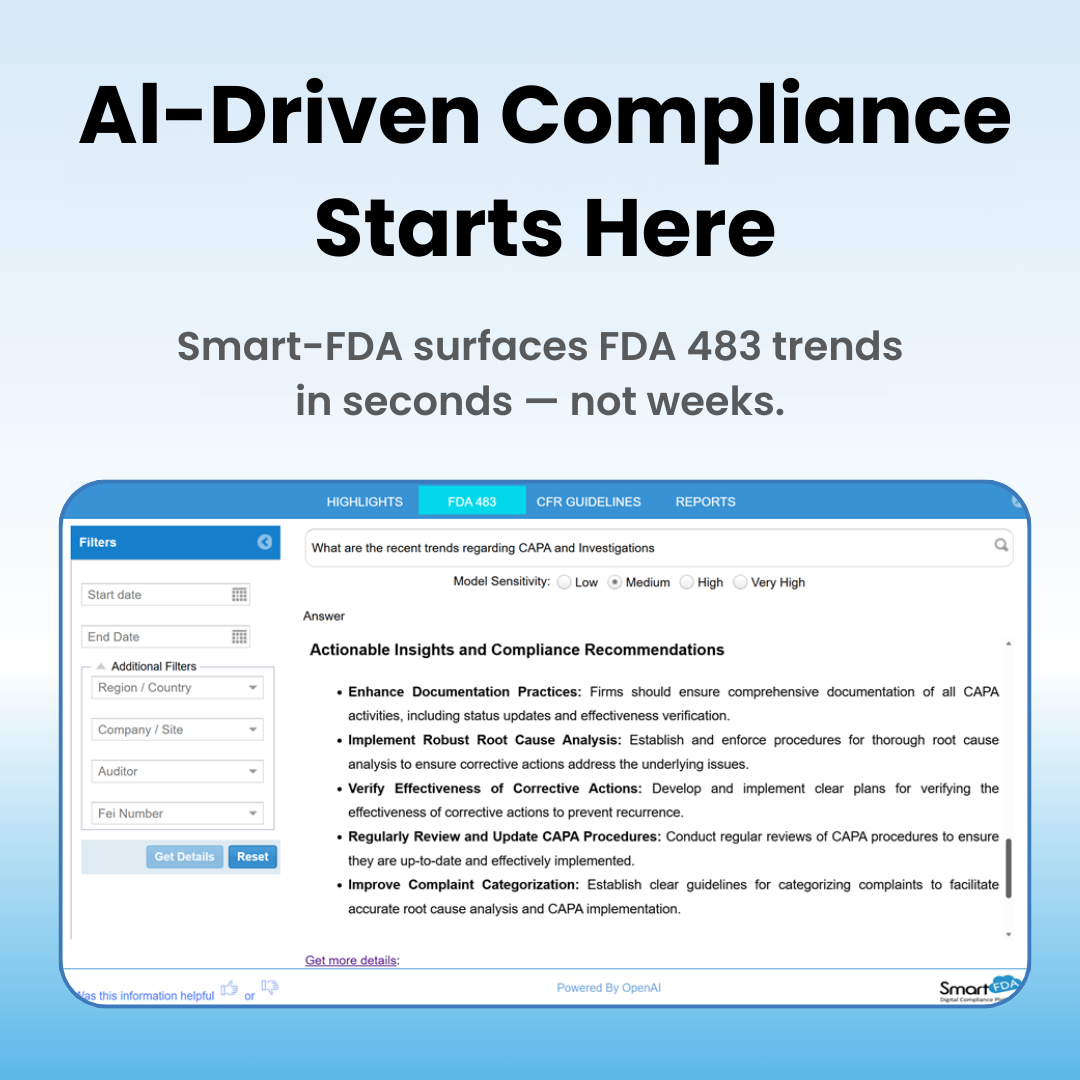
We recently asked a simple question: “What are the recent trends regarding CAPA and investigations?”
In seconds, Smart-FDA analyzed thousands of 483 observations and surfaced the most critical patterns.
Key CAPA FDA 483 Trends: Highlights
Here are a few key highlights:
- Inadequate documentation of CAPA activities
- Lack of Verification of Effectiveness (VoE)
- Failure to conduct proper root cause analysis
- Incomplete or inadequate CAPA procedures
- Inconsistent categorization and analysis of complaints
Regulatory Implications
- Potential non-compliance with FDA regulations, which can lead to warning letters or enforcement actions
- Increased risk of recurring issues due to missing root cause analysis and ineffective CAPA verification
- Possible delays in product approvals due to weak CAPA processes
The Power of AI-Driven Compliance
This is the power of AI-driven compliance:
- Faster insights
- Broader access to regulatory intelligence
- Smarter decisions across your quality and compliance teams
To see what Smart-FDA can surface for your organization, contact us and send us your question.
