AI in FDA Compliance: 7 Powerful Ways Smart-FDA Is Revolutionizing Regulatory Success
AI in FDA compliance is transforming how regulated industries manage inspections, interpret findings, and maintain operational continuity. In highly regulated sectors such as pharmaceuticals, medical devices, and biotechnology, compliance is complex, resource-intensive, and constantly evolving. Organizations must monitor regulatory trends, interpret inspection findings, and respond quickly to stay ahead of regulatory risk.
With the launch of Smart-FDA, we are applying AI in FDA compliance directly to one of the most challenging areas of regulation: understanding and acting on FDA inspection observations.
1. Making FDA 483 Observations Accessible Across the Organization
FDA Form 483 observations contain critical insights into compliance gaps identified during inspections. Traditionally, interpreting these observations requires specialized regulatory expertise, which limits access to a small group of compliance professionals.
Smart-FDA changes this dynamic. The platform provides pre-configured AI in FDA compliance summaries tailored to different roles, including:
- Quality Assurance leaders
- Regulatory affairs teams
- Manufacturing management
- Laboratory personnel
- Executive stakeholders
By translating complex regulatory language into clear, role-specific insights, Smart-FDA enables broader organizational awareness and faster decision-making.
2. Role-Based AI Insights for Faster Decisions
Compliance decisions often stall because relevant information is difficult to interpret or distribute. Smart-FDA addresses this by delivering concise, actionable summaries that highlight:
- Key risks and trends
- Root causes of common observations
- Potential operational impacts
- Recommended focus areas
This ensures that decision-makers receive the information they need without having to sift through lengthy reports or technical documentation.
3. Exploring Specific Compliance Questions with AI in FDA Compliance
Compliance challenges are rarely generic. Organizations frequently need insight into highly specific topics relevant to their operations.
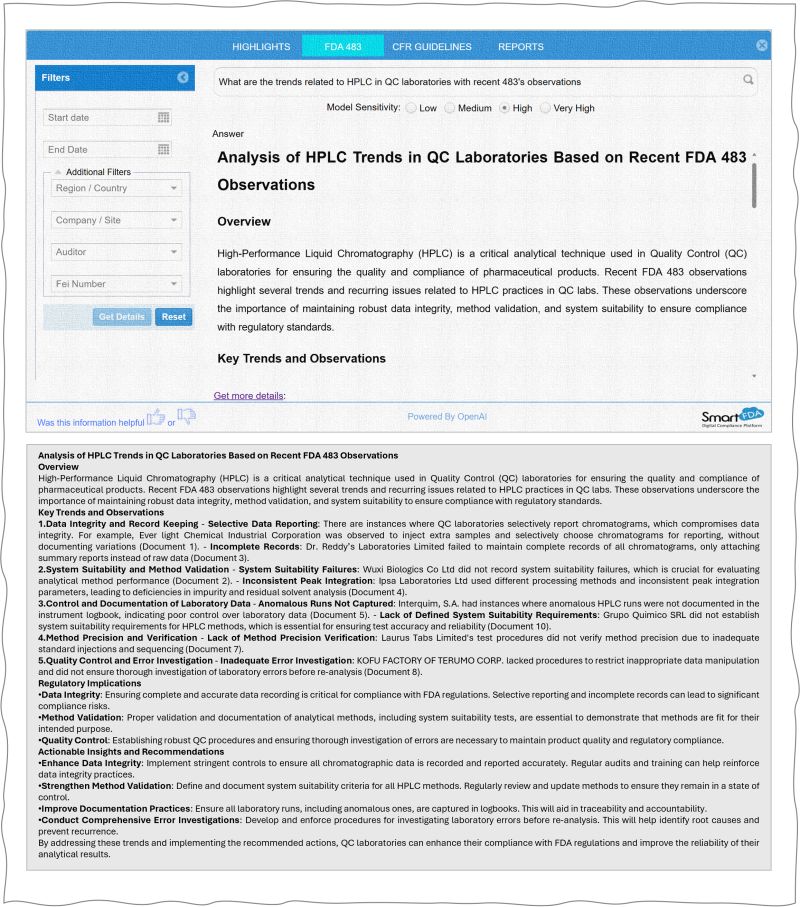
For example, a Quality Control laboratory may want to understand emerging regulatory concerns related to High-Performance Liquid Chromatography (HPLC) systems. Instead of manually reviewing numerous inspection reports, users can simply ask Smart-FDA targeted questions such as:
“What trends are emerging in FDA observations related to HPLC in QC laboratories?”
Within seconds, the platform generates a focused summary identifying:
- Recurring deficiencies
- Documentation issues
- Method validation concerns
- Data integrity risks
- Preventive measures observed across the industry
This capability allows teams to proactively address vulnerabilities before they become compliance findings.
4. Turning Regulatory Data into Strategic Intelligence
Beyond answering individual questions, Smart-FDA transforms historical inspection data into forward-looking intelligence. Organizations leveraging AI in FDA compliance can use these insights to:
- Strengthen internal audit programs
- Prioritize remediation efforts
- Improve inspection readiness
- Allocate resources more effectively
- Reduce the likelihood of repeat observations
Instead of reacting to compliance issues after inspections occur, companies can adopt a preventive approach driven by real regulatory intelligence.
5. Practical AI for Real-World FDA Compliance Needs
Many AI initiatives fail because they prioritize novelty over usability. Smart-FDA focuses on practical outcomes: helping organizations understand regulatory expectations and act on them efficiently.
Key characteristics of applied AI in FDA compliance include:
- Context-aware analysis of regulatory data
- Clear, non-technical summaries
- Rapid response times
- Relevance to specific operational functions
- Support for risk-based decision making
This approach ensures that AI serves as a decision support tool rather than a theoretical experiment.
6. Supporting a Culture of Compliance Across the Organization
Effective compliance requires organization-wide engagement, not isolated expertise. By making AI in FDA compliance insights accessible to multiple stakeholders, Smart-FDA helps foster a proactive compliance culture where risks are identified early and addressed collaboratively.
Teams can align on priorities, leadership gains visibility into emerging risks, and operational units understand how their activities influence regulatory outcomes.
7. The Future of AI in FDA Compliance
As regulatory scrutiny intensifies and data volumes continue to grow, AI in FDA compliance will become essential for maintaining efficiency. Solutions like Smart-FDA demonstrate how advanced analytics can be deployed responsibly to enhance — not replace — human expertise.
Organizations that adopt intelligent compliance tools early will be better positioned to navigate evolving regulations while maintaining quality, safety, and operational resilience.
Learn More About Smart-FDA
Smart-FDA represents a powerful step forward in applying AI in FDA compliance to regulatory challenges. By delivering targeted insights, role-based summaries, and rapid analysis of FDA 483 observations, the platform helps organizations move from reactive compliance management to proactive risk mitigation.
Contact us to learn more about the Smart-FDA AI-driven compliance platform and how it can support your organization’s regulatory strategy.
