AI is transforming compliance – and we’re leading the way.
With the launch of Smart-FDA, we’re putting FDA 483 intelligence in the hands of every stakeholder across your organization, not just a small circle of compliance experts.
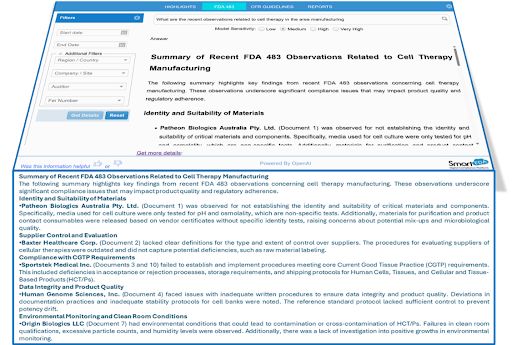
We put it to the test with a simple question: “What are the recent observations related to cell therapy in the area of manufacturing?”
In seconds, Smart-FDA surfaced the most critical trends:
- Identity and Suitability of Materials
- Supplier Control and Evaluation
- Compliance with CGTP Requirements
- Data Integrity and Product Quality
- Environmental Monitoring and Clean Room Conditions
- Laboratory Controls and Specifications
- Quality Program Implementation
- Implications for Regulatory Compliance
That’s the power of AI-driven compliance – faster insights, broader access, and smarter decisions across your entire team.
Organizations looking to surface FDA 483 trends faster are welcome to contact us at Smart-FDA to learn what AI-driven Smart-FDA compliance can do for your team.
