AI Compliance Trends Reshaping Regulatory Intelligence
AI compliance trends are transforming how regulated organizations interpret FDA 483 observations, manage inspection risk, and strengthen enterprise-wide quality systems. Instead of relying solely on manual document review and isolated expertise, organizations are now leveraging artificial intelligence to generate structured regulatory intelligence in seconds.
In regulated environments such as pharmaceuticals, biotechnology, and advanced manufacturing, inspection observations often reveal recurring compliance themes. Without centralized analysis, these patterns remain fragmented across multiple reports and sites. AI compliance trends solve this problem by converting unstructured regulatory findings into searchable, categorized insight.
Why AI Compliance Trends Matter in 2026
Regulatory expectations continue to rise globally. Agencies expect data integrity, documented quality oversight, supplier transparency, and proactive risk management. AI compliance trends enable organizations to move from reactive compliance toward predictive oversight.
Key drivers behind adoption include:
- Increasing inspection complexity
- Expansion of cell and gene therapy manufacturing
- Greater emphasis on CGTP adherence
- Heightened focus on data governance
- Multi-site operational oversight challenges
By structuring historical inspection findings, AI systems allow compliance leaders to see patterns that would otherwise require weeks of manual review.
AI Compliance Trends in FDA 483 Analysis
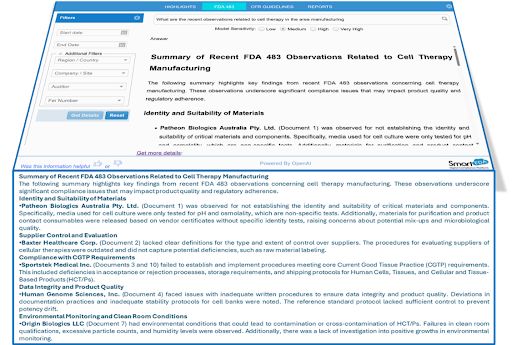
When evaluating recent inspection observations related to cell therapy manufacturing, AI-powered systems can rapidly cluster findings into recurring regulatory domains.
Common Inspection Themes Identified
- Identity and suitability of raw materials
- Supplier control and evaluation gaps
- Compliance with CGTP requirements
- Data integrity weaknesses
- Environmental monitoring deviations
- Cleanroom qualification inconsistencies
- Laboratory controls and specifications
- Quality system implementation gaps
AI compliance trends allow these inspection domains to be indexed, ranked, and cross-referenced across multiple years of FDA 483 data.
Instead of static document archives, organizations gain dynamic regulatory visibility.
Operational Benefits of AI Compliance Trends
The operational impact extends beyond document search functionality. AI compliance trends reshape how regulatory knowledge is shared and applied.
1. Accelerated Insight Generation
Manual inspection review processes can take days or weeks. AI-powered querying reduces this to seconds, allowing teams to focus on remediation rather than discovery.
2. Enterprise Accessibility
Regulatory intelligence is no longer limited to a small compliance circle. Manufacturing, quality, and executive leadership teams gain structured visibility into inspection trends.
3. Trend-Based CAPA Strategy
Corrective and Preventive Actions can be prioritized using historical recurrence data rather than isolated observations.
4. Improved Audit Readiness
By continuously monitoring inspection patterns, organizations strengthen inspection preparedness before regulators arrive.
Data-Driven Compliance Strategy
AI compliance trends introduce measurable consistency into compliance strategy. Rather than anecdotal interpretation, inspection findings are categorized using standardized criteria.
This enables:
- Cross-site inspection benchmarking
- Supplier performance comparison
- Trend mapping across product categories
- Longitudinal analysis of recurring regulatory gaps
Such capabilities improve governance transparency and support evidence-based decision-making.
Smart-FDA and AI Compliance
Smart-FDA operationalizes AI compliance trends by transforming FDA 483 intelligence into structured regulatory insight. Instead of manually reviewing observation narratives, users can search categorized themes and instantly retrieve related findings.
This structured approach supports:
- Manufacturing risk assessments
- Quality leadership reporting
- Supplier oversight reviews
- Regulatory strategy planning
By centralizing inspection intelligence, Smart-FDA enhances cross-functional coordination and reduces dependency on fragmented documentation systems.
The Future of AI Compliance Trends
The evolution of AI compliance trends will extend into predictive and prescriptive analytics. Emerging capabilities may include:
- Forecasting inspection focus areas based on historical data
- Automated regulatory gap detection
- Risk-weighted supplier evaluation models
- Integration with enterprise Quality Management Systems
As regulatory expectations continue to evolve, organizations equipped with AI-driven compliance intelligence gain strategic resilience.
AI compliance trends are not simply automation tools. They represent a structural shift toward data-informed regulatory governance, enterprise transparency, and continuous quality improvement.
If your organization is exploring AI compliance trends to strengthen FDA 483 intelligence and regulatory strategy, contact us to learn how smart-fda can support your compliance objectives.
