Scheduling Complexity in the Lab: A Different Scale of Challenge
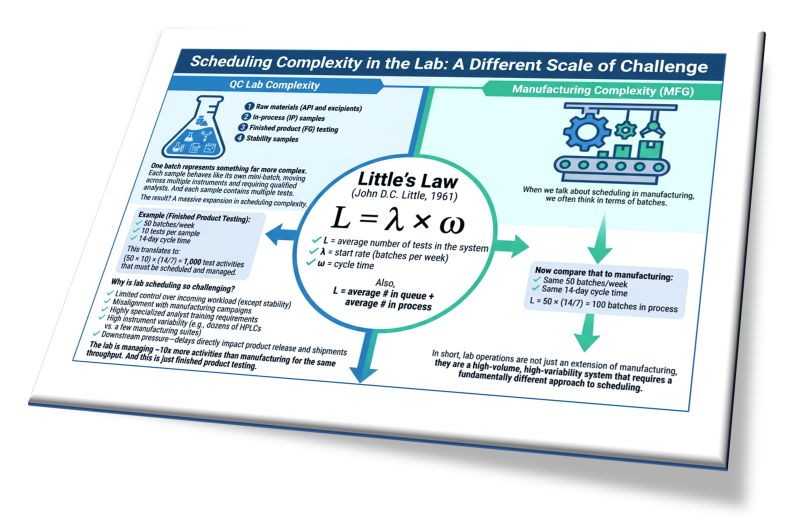
When we talk about scheduling in manufacturing, we often think in terms of batches. But in the lab, a single batch represents something far more complex.
One batch can include:
1️⃣ Raw materials (API and excipients) requiring 5–20 tests
2️⃣ In-process (IP) samples
3️⃣ Finished product (FG) testing
4️⃣ Stability samples
Each sample behaves like its own mini-batch, moving across multiple instruments and requiring qualified analysts. And each sample contains multiple tests. The result? A massive expansion in scheduling complexity.
Let’s quantify that using Little’s Law (John D.C. Little, 1961):
L = λ × ω
Where:
✔ L = average number of tests in the system
✔ λ = start rate (batches per week)
✔ ω = cycle time
Example (Finished Product Testing):
✔ 50 batches/week
✔ 10 tests per sample
✔ 14-day cycle time
This translates to: (50 × 10) × (14/7) = 1,000 test activities that must be scheduled and managed.
Now compare that to manufacturing:
✔ Same 50 batches/week
✔ Same 14-day cycle time
L = 50 × (14/7) = 100 batches in process
The lab is managing ~10x more activities than manufacturing for the same throughput. And this is just finished product testing. When you add raw materials, in-process testing, and stability programs, the complexity increases even further.
Why is lab scheduling so challenging?
✔ Limited control over incoming workload (except stability)
✔ Misalignment with manufacturing campaigns
✔ Highly specialized analyst training requirements
✔ High instrument variability (e.g., dozens of HPLCs vs. a few manufacturing suites)
✔ Downstream pressure—delays directly impact product release and shipments
In short, lab operations are not just an extension of manufacturing, they are a high-volume, high-variability system that requires a fundamentally different approach to scheduling.
Lab scheduling complexity is often underestimated. But in reality, it’s one of the most critical and challenging elements in ensuring supply chain performance.
